/PeriodicTableCharge-BW-56a12db13df78cf772682c34.png)
Examplesįor example, sodium is in group 1, indicating it has one valence electron. In this sense, groups 1 and 2 in the diagram below stay the same, but group 13 is our new “group 3”, group 14 is our new “group 4,” and so on. They are located in the block in the middle of the periodic table. This also means that when looking at a group number, exclude the transition metals. Thus you should take a look at the element’s specific electron shell configuration to figure it out. 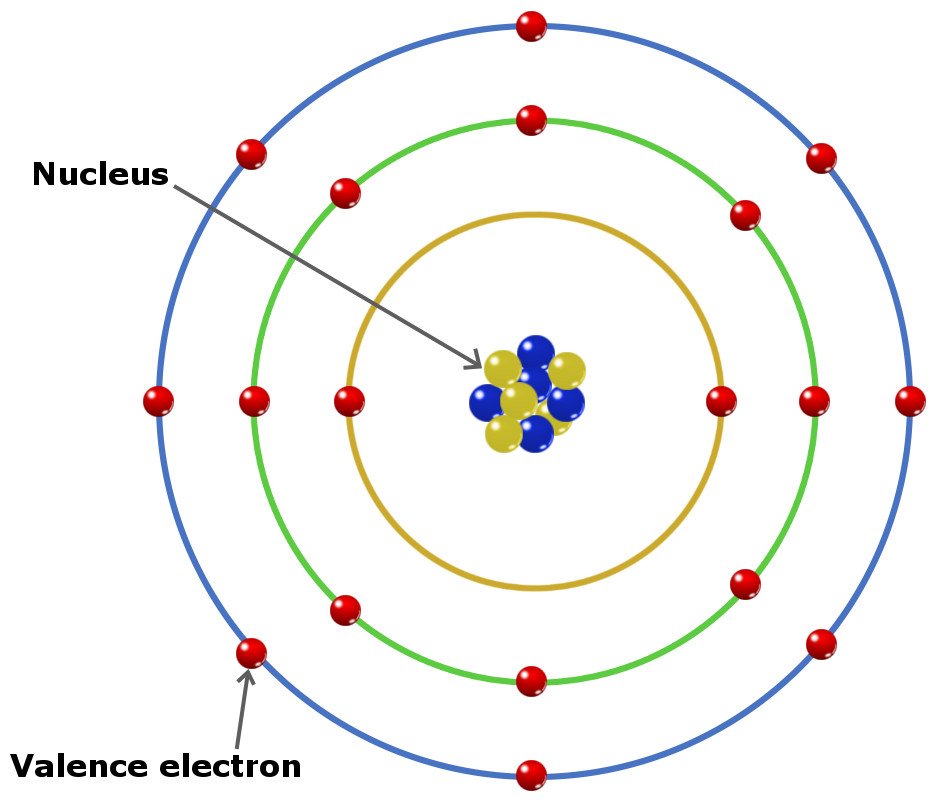
Transition metals have more complicated electron configurations. Note, however, that this rule only applies to elements that are not transition metals. Look at the group that the element is in, as the group number indicates the number of valence electrons that the element has. You can use the periodic table to help you determine how many valence electrons an element (specifically, a neutral atom of the element) has. How many valence electrons does an element have? On the other hand, nitrogen can form NH 3 so it has a valence of 3, and 3 valence electrons. An element’s valence was historically determined by how many hydrogen atoms it could bond to (which is determined by how many valence electrons it has available for bonding): for example, carbon can form CH 4 so it has a valence of 4, and 4 valence electrons. The term valence refers to the ability of an element to form bonds with other atoms. It is located on the outermost shell (in this case, the shell resembles a ring). This also means that the number of valence electrons that an element has determines its reactivity, electronegativity, and the number of bonds it can form.įor example, in the figure below showing a simplified diagram of sodium’s electron configuration, the valence electron is shown in red. These electrons, being the furthest from the nucleus and thus the least tightly held by the atom, are the electrons that participate in bonds and reactions. Valence electrons are electrons that located in the outermost electron shell of an atom. What are valence electrons? Why are they significant? We will also learn how to tell how many valence electrons an element has. In this tutorial, we learn about valence electrons, what they are, and why they are significant.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
